AS Problem Collection¶
| w23 | 11 | x | 21 | |
|---|---|---|---|---|
| 12 | x | 22 | ||
| 13 | 23 | |||
| w22 | 11 | x | 21 | |
| 12 | x | 22 | ||
| 13 | x | 23 | ||
| w21 | 11 | x | 21 | |
| 12 | x | 22 | ||
| 13 | 23 |
9701_w22_qp_12¶
18¶
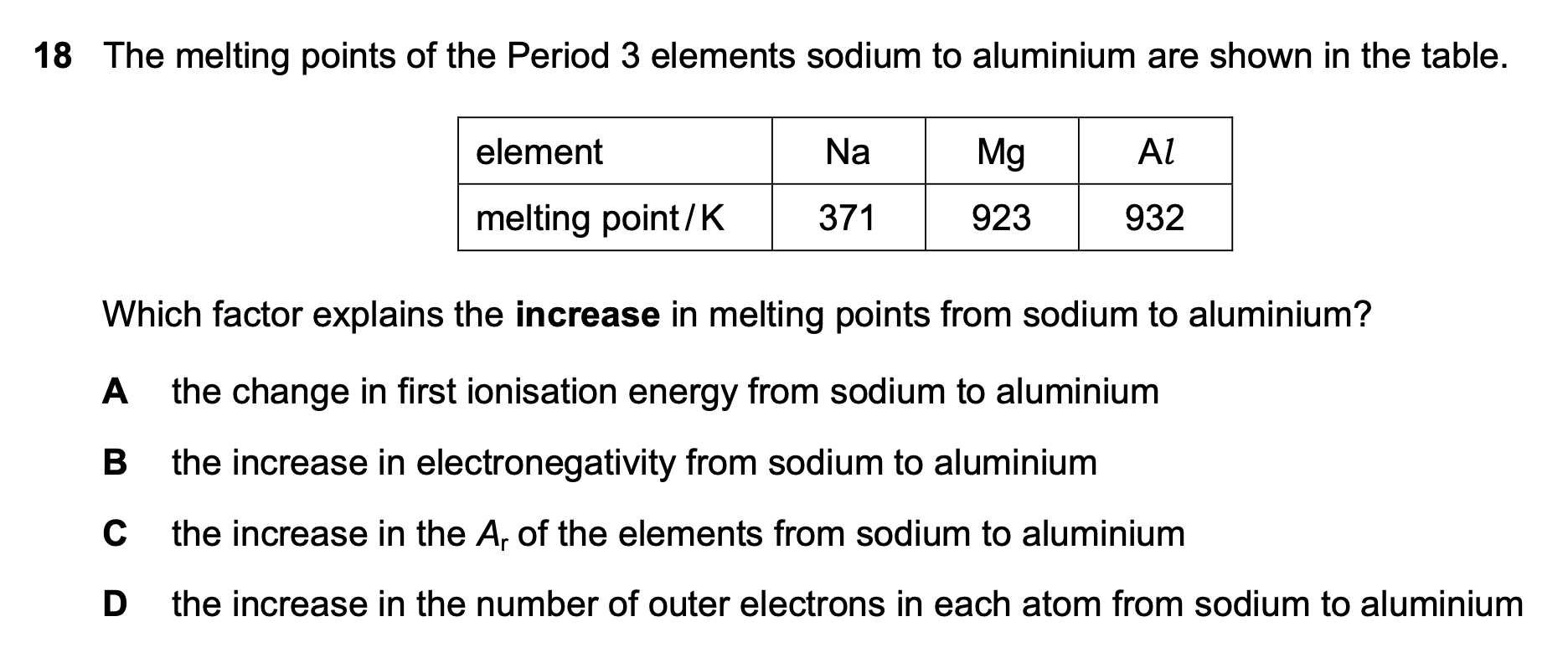
| Wrong Ans | C |
|---|---|
| Correct Ans | D |
| Explain | The strength of metal bonds is decided by the number of delocalized electrons |
9701_w21_qp_12¶
11¶

| Wrong Ans | B |
|---|---|
| Correct Ans | D |
| Explain | Both are catalysts |
16¶

| Wrong Ans | D |
|---|---|
| Correct Ans | C |
| Explain | dissolves in ammonia to form ; does not solve in ammonia |
26¶

| Wrong Ans | B |
|---|---|
| Correct Ans | D |
| Explain | Alcohol's corresponding carboxylic acid has stronger H-bond than itself. Aldehydes and ketones do not have H-bonds. |
29¶

| Wrong Ans | A |
|---|---|
| Correct Ans | B |
| Explain | only react with |
31¶
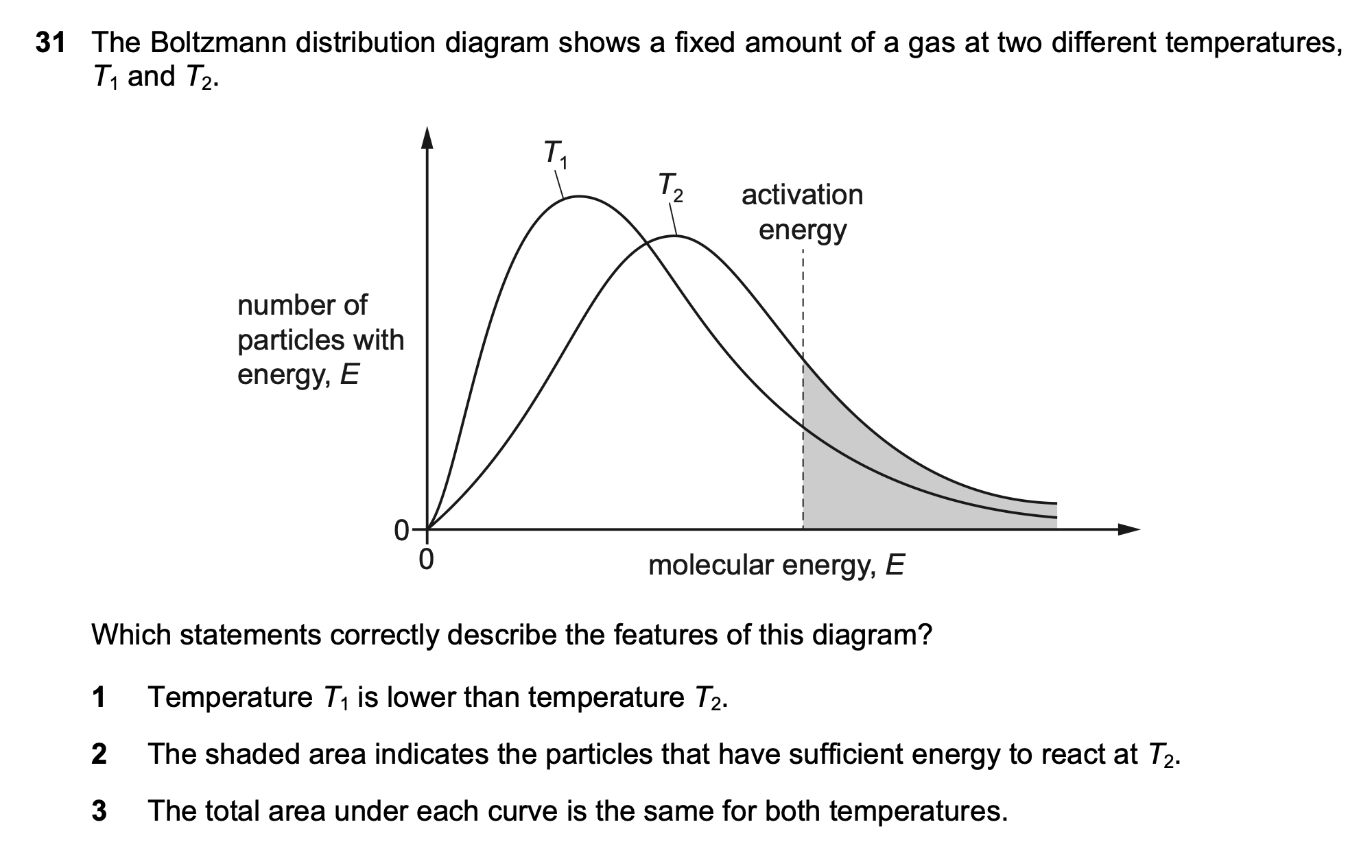
| Wrong Ans | 1, 2 |
|---|---|
| Correct Ans | 1, 2, 3 |
| Explain | The total area represents the total number of particles |
9701_w23_qp_11¶
5¶
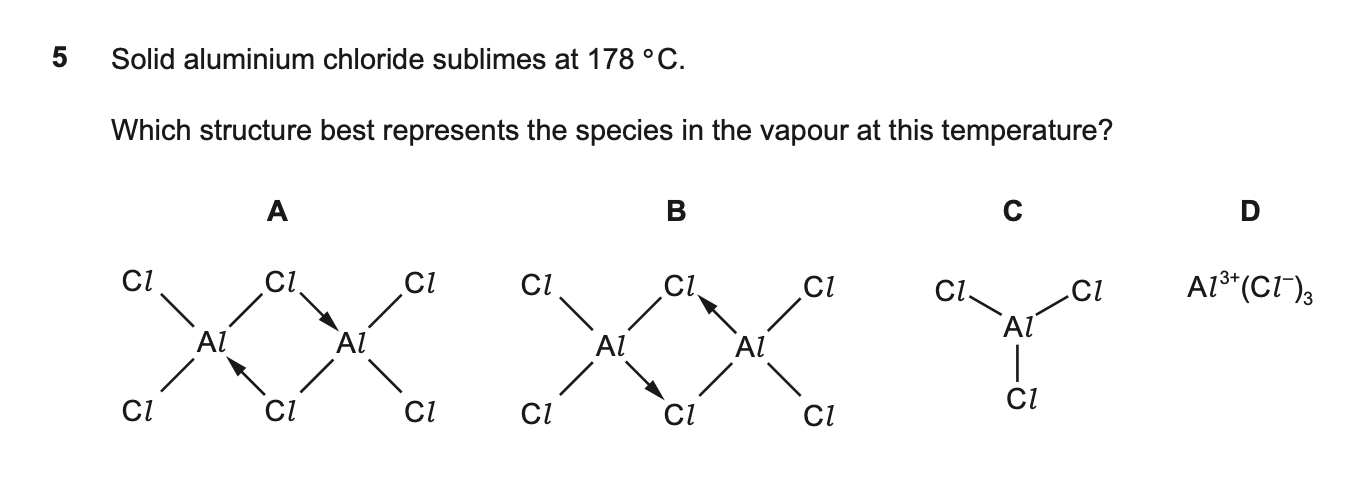
| Wrong Ans | C |
|---|---|
| Correct Ans | A |
| Explain | As solid, forms giant lattice. As liquid and vapor (< 400℃), it forms dimer. As vapor (> 400℃), it forms monomer. |
16¶

| Wrong Ans | B |
|---|---|
| Correct Ans | A |
| Explain |
Extra:
9701_w22_qp_11¶
Ground state: lowest possible energy state
2NaBr+2H2SO4→Br2+SO2+Na2SO4+2H2O
29¶
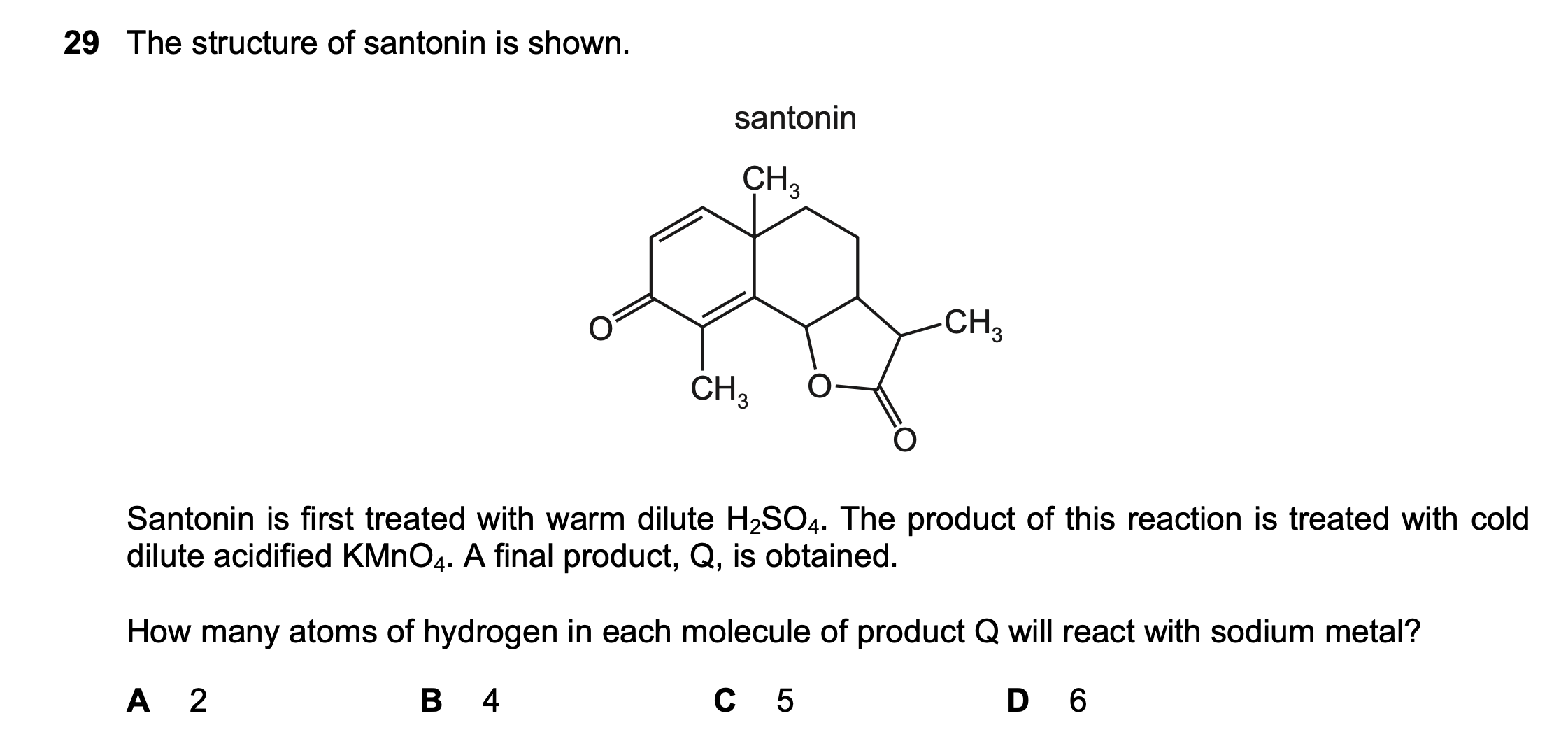
| Wrong Ans | A |
|---|---|
| Correct Ans | D |
| Explain | opens of the bottom right ring; add OH to each end of . does not provide O so alcohol is not oxidized to aldehyde or ketone, but can |
32¶
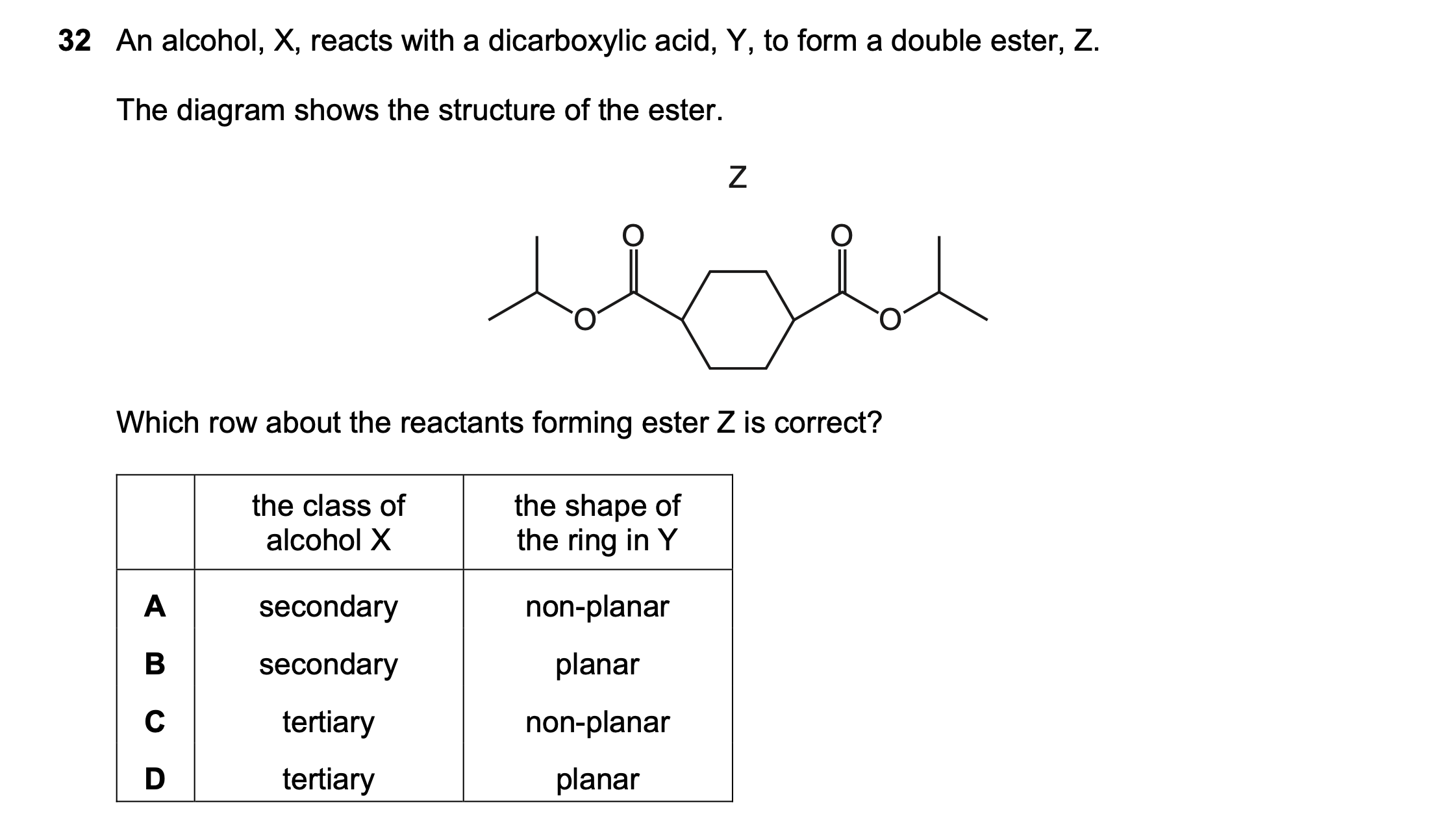
| Wrong Ans | B |
|---|---|
| Correct Ans | A |
| Explain | Saturated hexane ring is not planar but twisted. Benzane ring is planar. |
9701_w21_qp_11¶
8¶

| Wrong Ans | A |
|---|---|
| Correct Ans | C |
| Explain | Enthalpy change is analyzed under room condition (298 K + 100kPa) |
19¶

| Wrong Ans | D |
|---|---|
| Correct Ans | B |
| Explain | Does not directly linked to the formation photochemical smog. Though, + Volatile Organic Compounds photochemical smog. |
26¶

| Wrong Ans | C |
|---|---|
| Correct Ans | D |
| Explain | The intermediate is , with no . |
9701_w23_qp_12¶
17¶
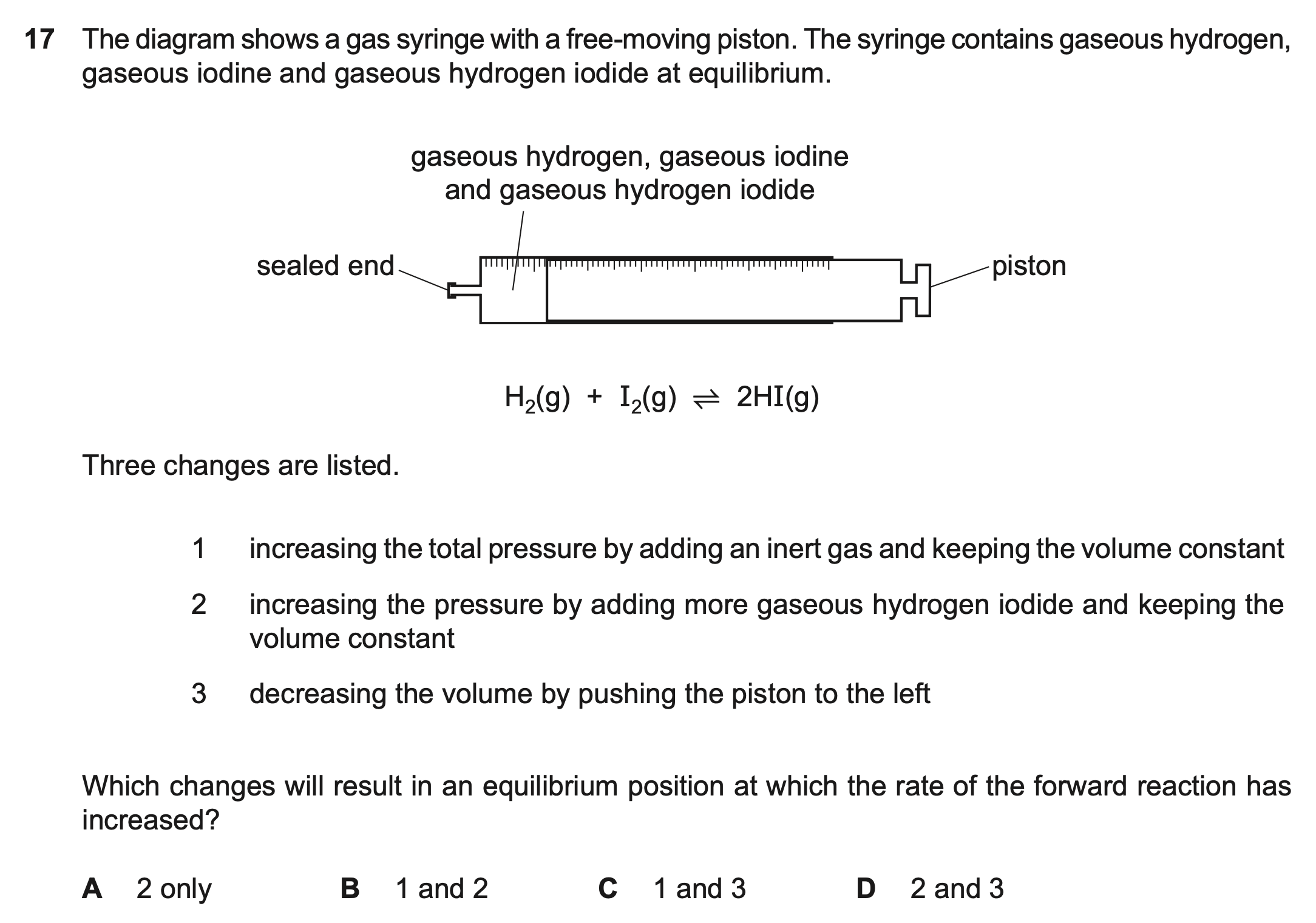
| Wrong Ans | X |
|---|---|
| Correct Ans | 2, 3 |
| Explain | For 2 and 3, though the equilibrium moves to the left/does not move, the concentration increases, so the rate of change of the forward reaction increases. |
33¶

| Wrong Ans | B |
|---|---|
| Correct Ans | A |
| Explain | NaOH(ethanol) can only be used in dehydrohalogenation, removing HX. To dehydrate, use conc. H₂SO₄. |